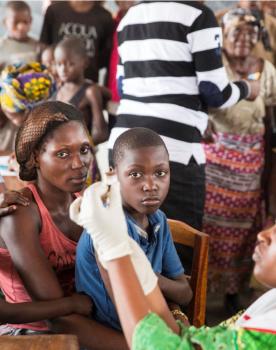Antibiogo
In brief
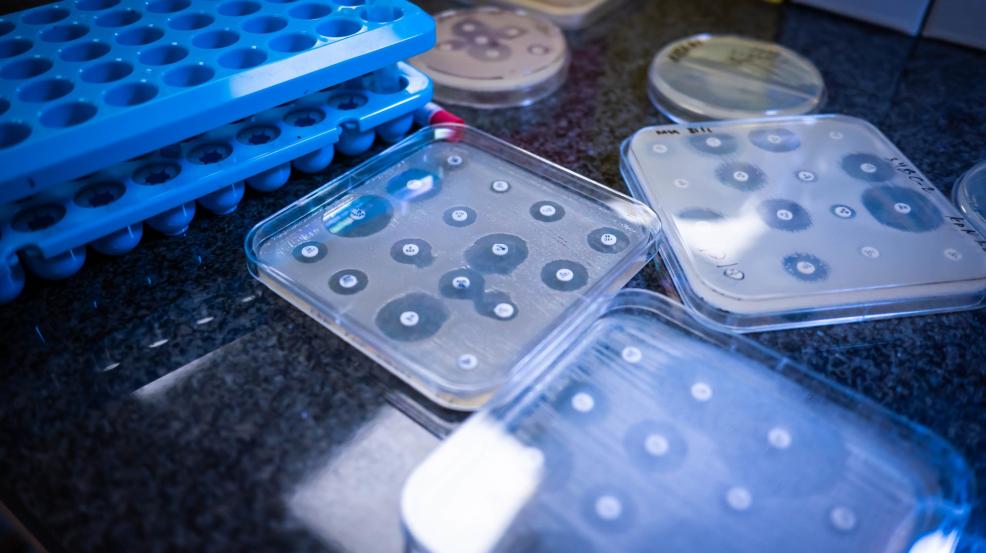
Antibiogo is a diagnostic aid medical device that aims to help doctors prescribe the most effective antibiotics to their patients. It is available as a free, open source and offline Android application. It allows non-expert laboratory technicians to measure and interpret antibiograms. It provides accurate results that can also be used for monitoring purposes and updating empirical treatments based on actual etiology.
Status of the project
News
In detail
Antibiogo fights against the spread of antimicrobial resistance and aims to guarantee equal access to a quality diagnosis, even in countries with limited resources where access cannot be assured today.

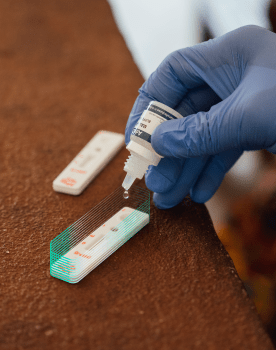
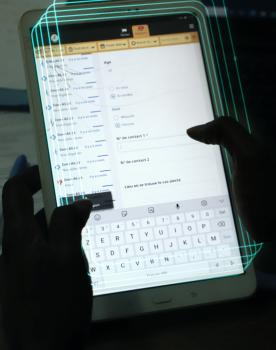
Antibiogo is a free smartphone application, available offline and open source (except for the expert system). It allows lab technicians to measure inhibition zone diameters found on antibiograms and interpret the results, without necessarily having expertise in microbiology, something which was previously required. The idea is not to replace microbiologists but to provide the necessary analytical support in countries where there is no access to these highly qualified resources.
In doing so, Antibiogo will improve the quality of the tests carried out in existing laboratories and will complement the establishment of quality bacteriology laboratories, even in the absence of an expert microbiologist in MSF intervention and more broadly in precarious contexts.
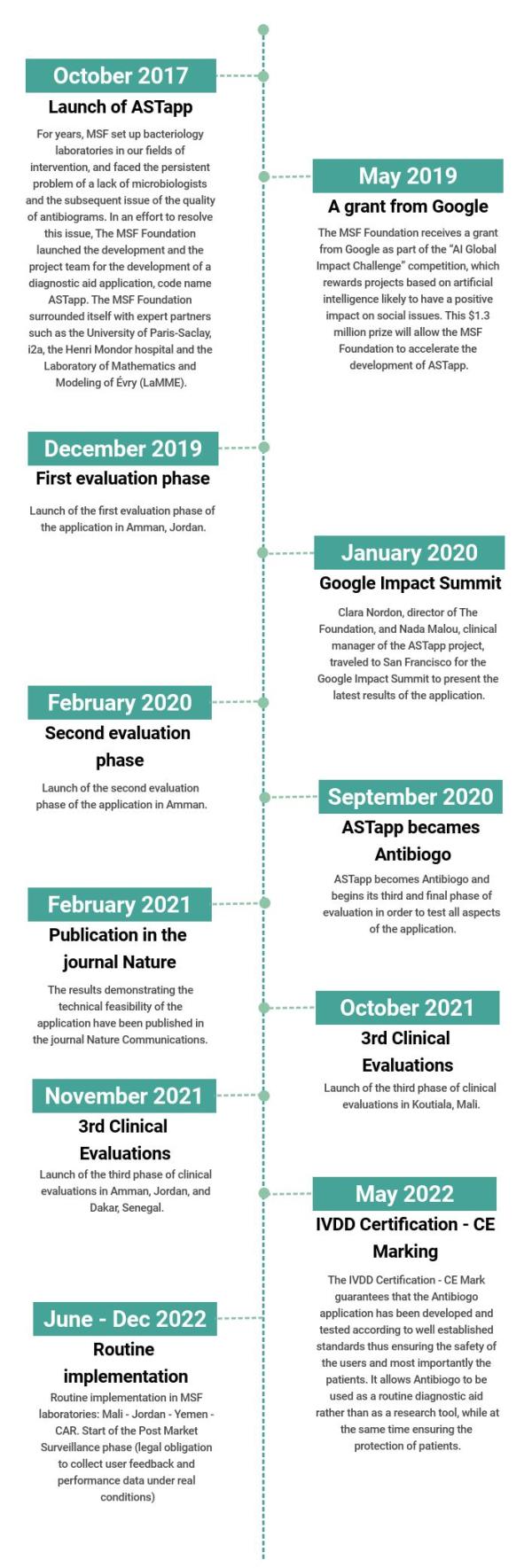
The MSF Foundation has been working on the project since 2017, in collaboration with external partners (technical, academic, financial) that it has been able to mobilize to reach today the certification, a major step for the deployment in LMIC.
The IVDD-CE marking is a guarantee that the Antibiogo application has been developed and tested according to well-established standards in order to ensure the safety of the users but especially of the patients.
It allows the use of Antibiogo in ICLs as a routine diagnostic aid and not in research mode while ensuring the protection of patients.
Antibiogo is the first CE-marked, in vitro medical device designed by MSF Foundation. This tool, which is a free downloadable app, provides a concrete response to slowing AMR in all countries by facilitating access to quality bacteriological diagnosis. The results can also be used for monitoring purposes and to update empirical treatment based on actual etiology.
The Antibiogo app is being implemented in several MSF laboratories, including Jordan and Kinshasa DRC and by December in Mali, the Central African Republic and Yemen and is intended to be deployed more extensively. With time, the app will be a tool for healthcare professionals in all LMICs.
Start of the Post Market Surveillance phase (legal obligation to collect user feedback and performance data under real conditions)
Launch of the final clinical evaluation in Mali

How does it work?
MSF uses AI to help treat patients and fight antibiotic resistance

What were the main stages of the project ?
Antibiogo in pictures
Our partners

Supporting the project
You wish to specifically support the development of this project? Contact Catherine Béchereau - Loyalty and Philanthropy Manager 01 40 21 56 88 - [email protected]